Why Hormone Health Matters to Orthopaedics—Without Expanding Orthopaedic Scope
by Fabian Poletti, MD, MSc (Imperial) DIC, FRCS (Eng), FEBOT (T&O)
Orthopaedic and Truma Surgeon
Regenerative & Joint Preservation Medicine
Disclaimer
This article does not argue that orthopaedic surgeons should prescribe menopausal hormone therapy. It argues that the musculoskeletal consequences of hormonal decline during the menopause transition are clinically relevant to orthopaedic care, and that better collaboration with clinicians specialised in women’s hormonal health is justified. Any discussion of menopausal hormone therapy (MHT/HRT) refers to medically supervised, individualised care under established women’s health guidelines and risk stratification.
The central argument
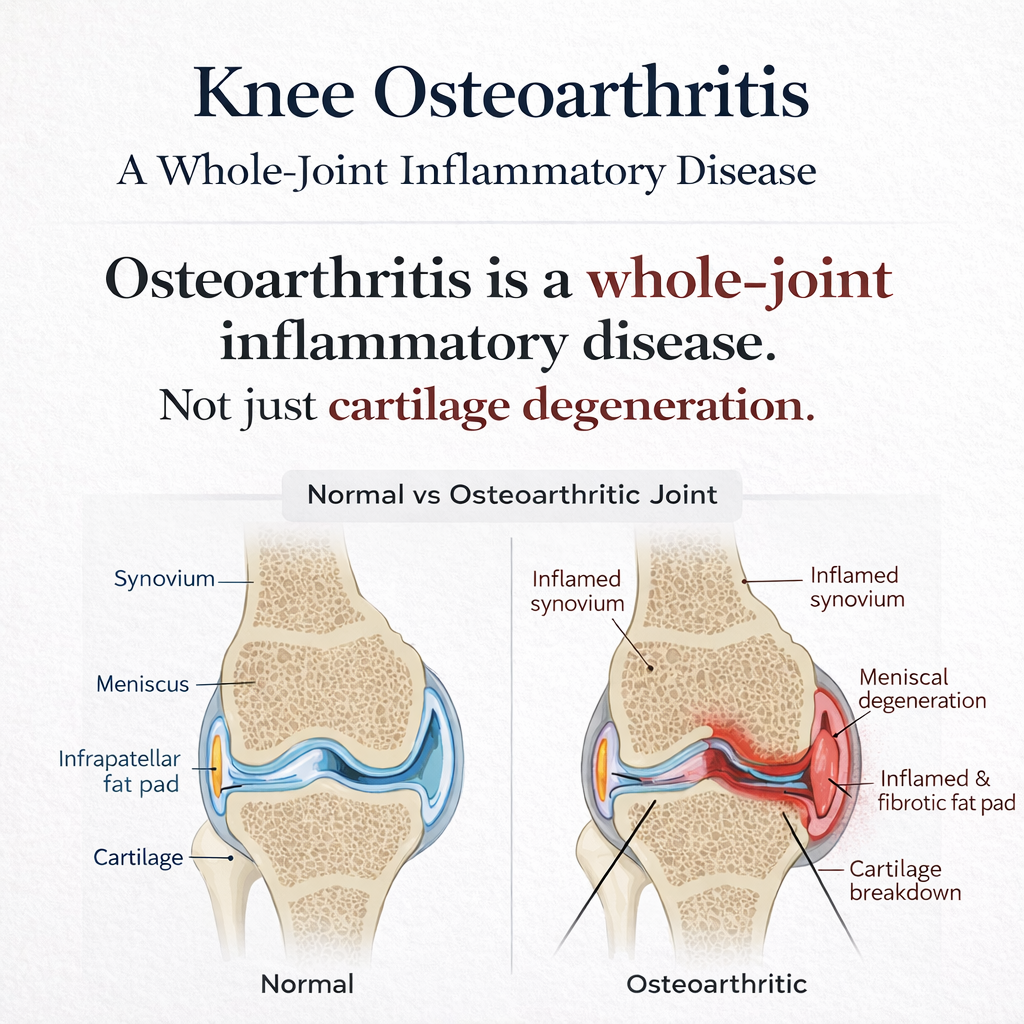
Knee osteoarthritis (OA) is still too often described as a simple “wear-and-tear” problem. That model is incomplete. Modern evidence supports OA as a whole-joint, immunometabolic, and neurosensory disease in which low-grade inflammation, subchondral remodelling, synovitis, adipose-derived mediators, muscle dysfunction, and pain sensitisation all influence symptoms and function.
This matters particularly in midlife women. The menopause transition is not just a reproductive milestone. It is a systemic endocrine shift that affects immune tone, connective tissue biology, muscle mass, sleep, recovery, bone, and pain processing. In practical terms, it may change the biological environment in which OA symptoms emerge, intensify, and respond to treatment.
The claim here is precise: menopause does not “cause” osteoarthritis de novo, and hormone therapy is not a cartilage-regenerating OA cure. But hormonal decline may amplify inflammatory tone, worsen pain sensitisation, reduce neuromuscular resilience, and impair recovery capacity in women with existing or developing joint disease. That makes it clinically relevant to orthopaedics.
Osteoarthritis is not just cartilage wear
OA is the most common chronic musculoskeletal disease and a major cause of pain, disability, reduced independence, healthcare use, and loss of physical function worldwide. Knee and hip OA matter most because they drive locomotor disability. When these joints fail, physical activity drops, muscle mass declines, weight often increases, metabolic risk worsens, and joint loading becomes more pathological. That creates a vicious cycle: reduced movement accelerates the same mechanical and biological conditions that worsen OA and push people toward joint replacement.
The old “wear-and-tear” model fails because clinical reality does not fit it cleanly. Orthopaedic practice constantly sees the paradox:
- some patients have severe radiographic degeneration with little pain
- others have major pain, stiffness, fatigue, and disability despite modest imaging findings
If OA pain were simply proportional to cartilage loss, that mismatch should not be so common. It exists because OA is a disease of the joint as an organ. It involves cartilage, yes, but also synovium, subchondral bone, menisci, ligaments, infrapatellar fat pad, periarticular muscles, and peripheral and central pain pathways.
Mechanical load still matters. But load acts inside a biological environment that can buffer stress or magnify damage.
Chronic low-grade inflammation: the silent amplifier
OA is increasingly recognised as a condition in which chronic low-grade inflammation amplifies tissue degeneration and pain. This is not the florid synovitis of classic inflammatory arthritis. It is subtler, slower, and easier to dismiss. But it is clinically important.
Key mediators include IL-6, TNF-α, and NF-κB, which influence:
- chondrocyte metabolism and matrix breakdown
- synovial activation
- subchondral bone remodelling
- nociceptor sensitisation and pain amplification
That biology fits what clinicians often see in midlife patients: pain not limited to loading, stiffness at rest, diffuse aching, fatigue, and poor recovery.
OA also interacts with systemic metabolism. Obesity, insulin resistance, dyslipidaemia, and adipose inflammation are not just background noise. Adipose tissue behaves as an endocrine organ, producing mediators that influence synovium, cartilage metabolism, and inflammatory tone. In knee OA, the infrapatellar fat pad is especially relevant; it is not just passive cushioning. It can secrete inflammatory and neuroactive substances that affect both local tissue biology and pain, sometimes independently of radiographic severity.
So the point is simple: pain in OA is not a pure readout of cartilage loss.
Pain is biological, not just structural
OA pain reflects both peripheral and central mechanisms. Peripheral nociceptors in synovium, subchondral bone, ligaments, and fat pad are sensitised by inflammatory mediators. Over time, repeated nociceptive input can alter central pain processing, leading to:
- lower pain thresholds
- broader pain distribution
- more persistent pain despite limited peripheral stimulus
This is why some patients with OA also present with sleep disturbance, fatigue, mood changes, and widespread pain features. Structural pathology and pain-amplification phenotypes frequently coexist. If you ignore that, you misread the patient.
Why the menopause transition matters
Perimenopause and menopause are often shoved into a gynaecological box. That is a mistake. They are systemic endocrine transitions with musculoskeletal consequences.
A 2026 systematic review and meta-analysis in JBJS Open Access, covering 37 studies and 93,021 women, found that muscle and joint pain increase significantly during perimenopause compared with premenopause, with risk around 1.35 times higher. Interestingly, symptom prevalence did not rise further from perimenopause to postmenopause, suggesting an inflection period rather than a slow, linear age-related process.
That finding matters, but it has limits. The review captured symptoms more reliably than orthopaedic diagnoses. It does not cleanly separate OA from tendinopathy, bursitis, myofascial pain, enthesopathy, or central sensitisation. So the evidence strongly supports a rise in musculoskeletal symptom burden during perimenopause, but diagnostic phenotyping remains weak.
Still, dismissing this as “just ageing” is no longer defensible.
Why women are disproportionately affected
Women make up a large proportion of OA cases globally and often experience greater symptom burden and structural deterioration after midlife. One plausible reason is that oestrogen receptors are present in cartilage, bone, synovium, muscle, and immune cells. Oestrogen signalling appears to influence inflammatory tone, tissue metabolism, and musculoskeletal resilience.
This does not mean oestrogen “prevents OA.” That would be sloppy and overstated. The more defensible claim is that endocrine change may modify the inflammatory and neuromuscular context in which OA symptoms and function evolve.

Hormonal decline as an amplifier, not a sole cause
The key distinction has to remain sharp: hormonal decline does not create OA from nothing. What it may do is shift the musculoskeletal system into a more vulnerable state.
1) Oestrogen
Oestrogen receptors are expressed across musculoskeletal tissues. At physiological levels, oestrogen is generally associated with:
- downregulation of NF-κB
- lower IL-6 and TNF-α signalling
- less pro-inflammatory immune behaviour
- effects on bone remodelling and osteoclast–osteoblast balance
Experimental oestrogen deficiency has been associated with cartilage degradation, chondrocyte dysfunction, and altered subchondral remodelling. That supports biological plausibility. But translating that directly into human structural OA progression is not straightforward.
Clinically, observational studies have suggested that oestrogen use may be associated with lower prevalence of radiographic knee or hip OA. But observational data are messy: confounding, healthcare access, BMI differences, activity patterns, and healthy-user bias all distort interpretation.
The strongest clinical signal is symptom relief, not structural reversal. Post hoc analyses from large trials such as the Women’s Health Initiative found modest but statistically significant reductions in joint pain among women receiving oestrogen versus placebo. The effect size was not huge, and OA structural progression was not the endpoint.
So the sober conclusion is this: oestrogen therapy may reduce joint pain in selected women; evidence for preventing or reversing structural OA remains limited and inconsistent.
2) Progesterone
Progesterone gets treated as if it only matters for reproduction. Wrong. It has broader neuroendocrine and immunomodulatory roles. It may affect cytokine signalling, microglial activity, nociception, and central nervous system regulation.
Clinically, progesterone decline is associated with sleep disturbance, increased pain sensitivity, and poorer recovery. That matters because sleep loss itself worsens inflammation and pain processing. Direct evidence linking progesterone replacement to OA structure is weak, but there is a plausible indirect pathway through improved sleep, pain modulation, and recovery quality.
So the evidence here is moderate for sleep and pain modulation, but low for OA-specific structural outcomes.
3) Testosterone
Women have testosterone too, and it matters. Physiological androgen levels help maintain lean mass, strength, power, and neuromuscular coordination. Decline in androgens may contribute to loss of muscle strength and dynamic stability.
That matters in knee OA because quadriceps weakness is consistently associated with pain severity and progression. If muscle strength declines, joint loading becomes less controlled and symptoms may worsen.
Trials of testosterone therapy in women have shown improvements in lean mass and strength under supervised care, but OA-specific outcomes are rarely studied directly. So again, plausible benefit through the muscle pathway, but no solid basis for claiming OA disease modification.
4) Vitamin D
Vitamin D is not a side note. It is a steroid hormone relevant to immune function, muscle performance, bone remodelling, and falls risk. Deficiency is common in midlife and older women. Some OA cohorts show associations between low vitamin D and greater pain, but structural trial results are mixed.
The most defensible position is that correcting deficiency may improve muscle function and fall risk, while cartilage-specific effects remain uncertain. Vitamin D optimisation should be treated as foundational musculoskeletal care, not a disease-specific OA cure.
What hormone therapy can and cannot do
This is where people start talking nonsense if you let them.
Menopausal hormone therapy does not:
- regrow cartilage
- reverse osteophytes
- correct malalignment
- eliminate established structural OA
Its possible role is more modest and more biologically coherent: it may help modify the disease environment.
When appropriately indicated, supervised, and individualised, MHT may:
- reduce chronic low-grade inflammatory tone
- modulate IL-6 and TNF-α signalling
- improve sleep and recovery
- improve muscle mass or neuromuscular resilience in some contexts
- reduce pain sensitisation
- improve response to rehabilitation
The evidence is best for symptoms and function, not for structural OA modification.
A reasonable evidence hierarchy looks like this:
- reduction in joint pain in selected women: moderate
- improvement in lean mass and strength, especially with androgen-relevant pathways: moderate
- reduction in falls risk through bone and muscle effects: moderate
- prevention or reversal of structural OA: limited and inconsistent
That distinction is non-negotiable if you want scientific credibility.
Timing matters
There is growing support for a timing hypothesis in menopausal hormone therapy: initiation closer to the menopause transition may produce more favourable systemic effects than late initiation. From an orthopaedic perspective, that fits the idea of a musculoskeletal inflection window.
If endocrine change influences inflammatory tone, pain thresholds, and neuromuscular resilience, earlier intervention during the transition may be more useful than trying to fix years of downstream catabolic adaptation later. That said, definitive OA-specific structural timing trials do not exist. So this remains biologically plausible, not proven.
Risk stratification and clinical boundaries
Hormone therapy is not for everyone. Any decision requires proper risk–benefit assessment by clinicians qualified in menopause care. Relevant considerations include:
- personal or high-risk history of hormone-sensitive malignancy
- thromboembolic risk
- cardiovascular profile
- age and years since menopause onset
- uterine status and endometrial protection needs
- migraine and vascular sensitivities
Modern menopause guidance emphasises individualisation, route, dose, timing, and monitoring. That is exactly why orthopaedics should not try to freeload into hormone prescribing. But pretending hormonal status is irrelevant is just as irresponsible.
What this means for orthopaedic practice
The practical implication is not scope creep. It is better phenotyping.
Endocrine-aware OA phenotyping
In midlife women, an endocrine-aware phenotype may include:
- pain disproportionate to radiographic severity
- abrupt symptom escalation over 1–3 years
- diffuse stiffness beyond the index joint
- sleep disturbance
- declining muscle strength without clear change in training load
- central sensitisation features
- menstrual irregularity or other menopause-transition symptoms
This does not replace structural assessment. It contextualises it.
Reinterpreting “treatment resistance”
A common scenario in midlife women with knee OA is:
- physiotherapy done
- injections done
- imaging-guided interventions done
- transient benefit, then recurrence
Too often this gets framed as poor compliance or inevitable degeneration. Sometimes the real problem is that the patient is being treated locally while living in a persistently pro-inflammatory, sleep-disrupted, neuromuscularly compromised biological state. Repeating local procedures in that environment may produce diminishing returns.
Structured referral, not orthopaedic overreach
Orthopaedic surgeons do not need to prescribe MHT. They do need to know when hormonal status may be clinically relevant and when to refer. That means:
- screening awareness for menstrual change, vasomotor symptoms, sleep disruption, unexplained weakness, changing pain phenotype
- defined referral pathways to menopause-informed gynaecologists, endocrinologists, or dedicated women’s health clinics
- shared outcome goals focused on pain, function, muscle preservation, recovery, and delay of surgery where appropriate
This is not ideological “integrative medicine.” It is systems-based clinical reasoning.
Implications for rehabilitation, joint preservation, and surgery
If the biological environment is hostile, rehabilitation works worse. So do injection-based and biologic strategies. A persistently inflamed, sleep-disrupted, sarcopenic patient is not the same rehabilitation substrate as one whose systemic physiology is optimised.
That means endocrine optimisation, when appropriate, may improve the durability of:
- strength rehabilitation
- load management
- weight optimisation
- regenerative or joint preservation strategies
The same logic applies to surgery. Arthroplasty corrects structure, but not systemic inflammatory tone, muscle loss, or central sensitisation. Those factors may contribute to slower recovery, persistent pain, and poorer functional gains even after technically successful surgery. Endocrine optimisation may prove relevant here too, although prospective outcome data are still limited.
What this model does not claim
To keep this honest, the boundaries have to stay explicit:
- hormone therapy does not reverse established cartilage loss
- hormone therapy is not indicated for every woman with OA
- biomechanics still matter enormously
- load management, weight control, and strength work remain foundational
- current evidence for structural disease modification is weak
- menopause-associated pain is not automatically OA and must be phenotyped properly
The argument is not “replace biomechanics with endocrinology.”
The argument is integrate biology with biomechanics.
Bottom line
OA in midlife women cannot be understood properly through imaging alone. The menopause transition appears to be a period of biological recalibration that affects immune tone, muscle, connective tissue, sleep, recovery, and pain processing. These changes do not single-handedly cause OA, but they may amplify symptoms, reduce resilience, and alter response to treatment.
The best current evidence supports moderate confidence that appropriately indicated menopausal hormone therapy may reduce joint pain and improve aspects of musculoskeletal function in selected women. Evidence that it modifies structural OA progression remains limited and inconsistent.
For orthopaedics, the message is pragmatic:
- assess the structure properly
- recognise the inflammatory and neuromuscular context
- consider endocrine transition in midlife women with disproportionate symptoms
- collaborate with menopause specialists when appropriate
Orthopaedics does not need to become endocrinology. But it does need to stop pretending hormonal biology is irrelevant.
That is not scope expansion. It is better medicine.
For readers interested in the full evidence synthesis and detailed scientific rationale, the complete preprint is available here:
https://www.preprints.org/manuscript/202603.0749
Leave a Reply